Selected Grade 2-4 Hepatic Laboratory Abnormalities and Adverse Drug Reactions in Treatment-Experienced Subjects Over 48 Weeks (Pooled DUET Analysis)
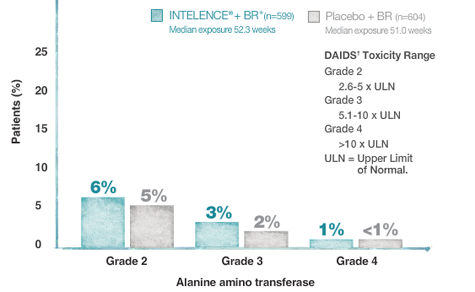
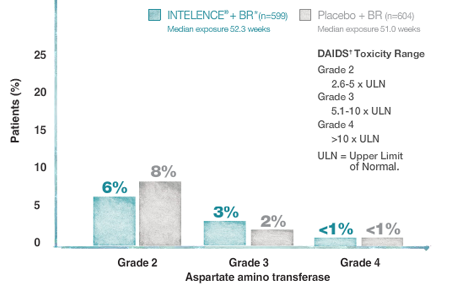
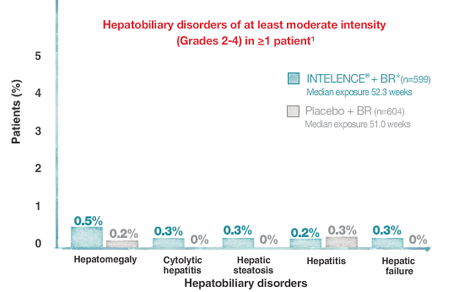
Please see full description of study design and baseline characteristics.
*The BR for INTELENCE® and placebo arms consisted of darunavir/ritonavir ± ≥2 other investigator-selected ARVs (N[t]RTIs ± ENF). In the INTELENCE® arm (n=599), 25.5% of patients used ENF de novo and 20.0% reused ENF. In the placebo arm (n=604), 26.5% of patients used ENF de novo and 20.4% reused ENF.
†Division of AIDS (DAIDS) of the National Institute of Allergy and Infectious Diseases and the National Cancer Institute (NCI).
