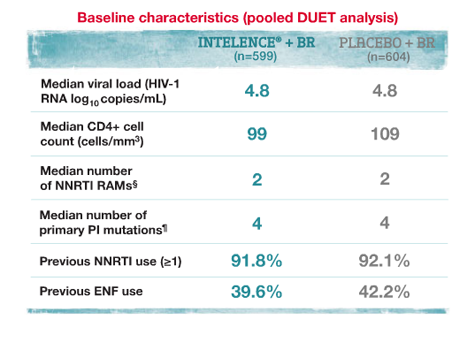
Virologic Response in Treatment-Experienced Adult Patients* With NNRTI and PI Resistance
Superior virologic response (<50 copies/mL) vs placebo + BR at Week 48
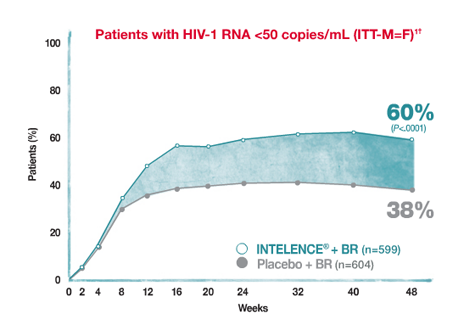
The BR for both arms consisted of darunavir/ritonavir + ≥2 other investigator-selected ARVs (N[t]RTIs ± ENF)‡
Rates of virologic failure (HIV-1 RNA ≥50 copies/mL) at Week 48 for overall INTELENCE® (etravirine) and placebo arms were 21% and 33%, respectively. Discontinuation rates before Week 48 were 10% and 18% due to virologic failure, 5% and 2% due to adverse events, and 3% and 5% due to other reasons, respectively.
Treatment Consideration
Use of INTELENCE® in combination with other active agents can increase the likelihood of treatment response. In patients who have had virologic failure on an NNRTI-containing regimen, INTELENCE® should not be used in combination with only N(t)RTIs.
- In a randomized, exploratory, open-label, Phase 2b trial (TMC 125-C227): treatment-experienced, PI-naïve patients with evidence of NNRTI resistance received 2 investigator-selected N(t)RTIs + either INTELENCE® (n=59) or an investigator-selected PI (n=57). Patients taking INTELENCE®, compared with control PI-treated patients, had lower antiviral responses associated with reduced susceptibility to the N(t)RTIs and INTELENCE®
Please see full description of study design and baseline characteristics.
*Treatment-experienced adult patients who had viral load (HIV-1 RNA) >5000 copies/mL, ≥1 NNRTI RAM (A98G, L100I, K101E/P/Q, K103H/N/S/T, V106A/M, V108I, E138G/K/Q, V179I/F/G, Y181C/I/V, Y188C/H/L, G190A/E/S, P225H, F227C, M230I/L, P236L, K238N/T, Y318F) at screening or from prior genotypic analysis, ≥3 primary PI mutations (D30N, V32I, L33F, M46I/L, I47A/V, G48V, I50L/V, V82A/F/L/S/T, I84V, N88S, or L90M) at screening, and were on a stable ARV regimen for ≥8 weeks.2
†Based on intent-to-treat (ITT) analysis missing equals failure (M=F).
‡In the INTELENCE® arm (n=599), 25.5% of patients used ENF de novo and 20.0% reused ENF. In the placebo arm (n=604), 26.5% of patients used ENF de novo and 20.4% reused ENF.